|
A New Drug Application for 68Ga-DOTATOC is being developed by the University of Iowa. 68Ga-DOTATATE (NETSPOT Advanced Accelerator Applications) is currently approved by the Food and Drug Administration. Newer imaging agents targeting SSTR labeled with 68Ga have subsequently been developed, namely, DOTATATE and DOTATOC ( 7). Representatives from the Society of Nuclear Medicine and Molecular Imaging (SNMMI), the American College of Radiology (ACR), the American Society of Clinical Oncology (ASCO), the North American Neuroendocrine Tumor Society (NANETS), the European Association of Nuclear Medicine (EANM), the Endocrine Society, the Society of Surgical Oncology, the National Comprehensive Cancer Network (NCCN), the American College of Physicians (ACP), the American Gastroenterological Association (AGA), and the World Conference on Interventional Oncology (WCIO) assembled under the auspices of an autonomous workgroup to develop the following AUC. Of the 12 clinical scenarios evaluated, 9 were graded as appropriate: initial staging after the histologic diagnosis of NET, evaluation of an unknown primary, evaluation of a mass suggestive of NET not amenable to endoscopic or percutaneous biopsy, staging of NET before planned surgery, monitoring of NET seen predominantly on SSTR PET, evaluation of patients with biochemical evidence and symptoms of a NET, evaluation of patients with biochemical evidence of a NET without evidence on CI or a prior histologic diagnosis, restaging at time of clinical or laboratory progression without progression on CI, and new indeterminate lesion on CI with unclear progression. The indications were evaluated in well-differentiated NETs. These appropriate use criteria (AUC) are intended to aid referring medical practitioners in the appropriate use of SSTR PET for imaging of patients with NETs. SSTR PET should replace 111In-pentetreotide scintigraphy (OctreoScan Mallinckrodt) in all indications in which the latter is currently being used. Results of the third study were published in the May 2016 issue of the Journal of Nuclear Medicine, and are the subject of an article published in the Applied Radiology Newsletter and posted online here.Somatostatin receptor (SSTR) PET has demonstrated a significant improvement over conventional imaging (CI) in patients with neuroendocrine tumors (NETs). The third demonstrated the safety and efficacy of the tracer for the diagnosis, staging, and treatment management of neuroendocrine tumors. 
The second evaluated 68Ga-Dotatate images using histopathology. The first compared 68Ga-Dotatate images of neuroendocrine tumors to images obtained with an approved drug, and then confirmed with CT or MRI. In its press release, the FDA referenced the three studies that established the safety and effectiveness of NETSPOT, all headed by researchers at Vanderbilt University Medical Center in Nashville, TN. It is currently approved for use with the GalliaPharm® Ga 68 generator from Eckert & Ziegler. An orphan drug is defined as a drug that treat diseases or conditions that affect 200,000 or fewer individuals in the United States or 5 out of 100,000 in the European Union.Īdvanced Accelerator Applications has announced that NETSPOT will be supplied in the United States by Cardinal Health of Dublin, OH, a healthcare services company operating the largest radiopharmaceutical network in the nation. Gallium 68Ga-Dotatate received Orphan Drug Designation in March 2014 from both the FDA and the European Medicines Agency. The uptake of 68Ga-Dotatate reflects the level of somatostatin receptor density in neuroendocrine tumors. 68Ga-Dotatate, a positron emitting analogue of somatostatin, works by binding to such receptors. Neuroendocrine tumors have receptors for somatostatin, a hormone that regulates the endocrine system. The tumors develop in the hormone-producing cells of the body’s neuroendocrine system, and are located in organs such as the stomach, intestines, pancreas, and lungs. Neuroendocrine tumors are malignant and benign tumors that affect an estimated 47,300 individuals in Europe and the United States each year. The commercial product, a single dose kit for the preparation of a 68Ga-Dotatate injection manufactured by Advanced Accelerator Applications of Saint Genis-Pouilly, France, has been given the market name NETSPOT ™. 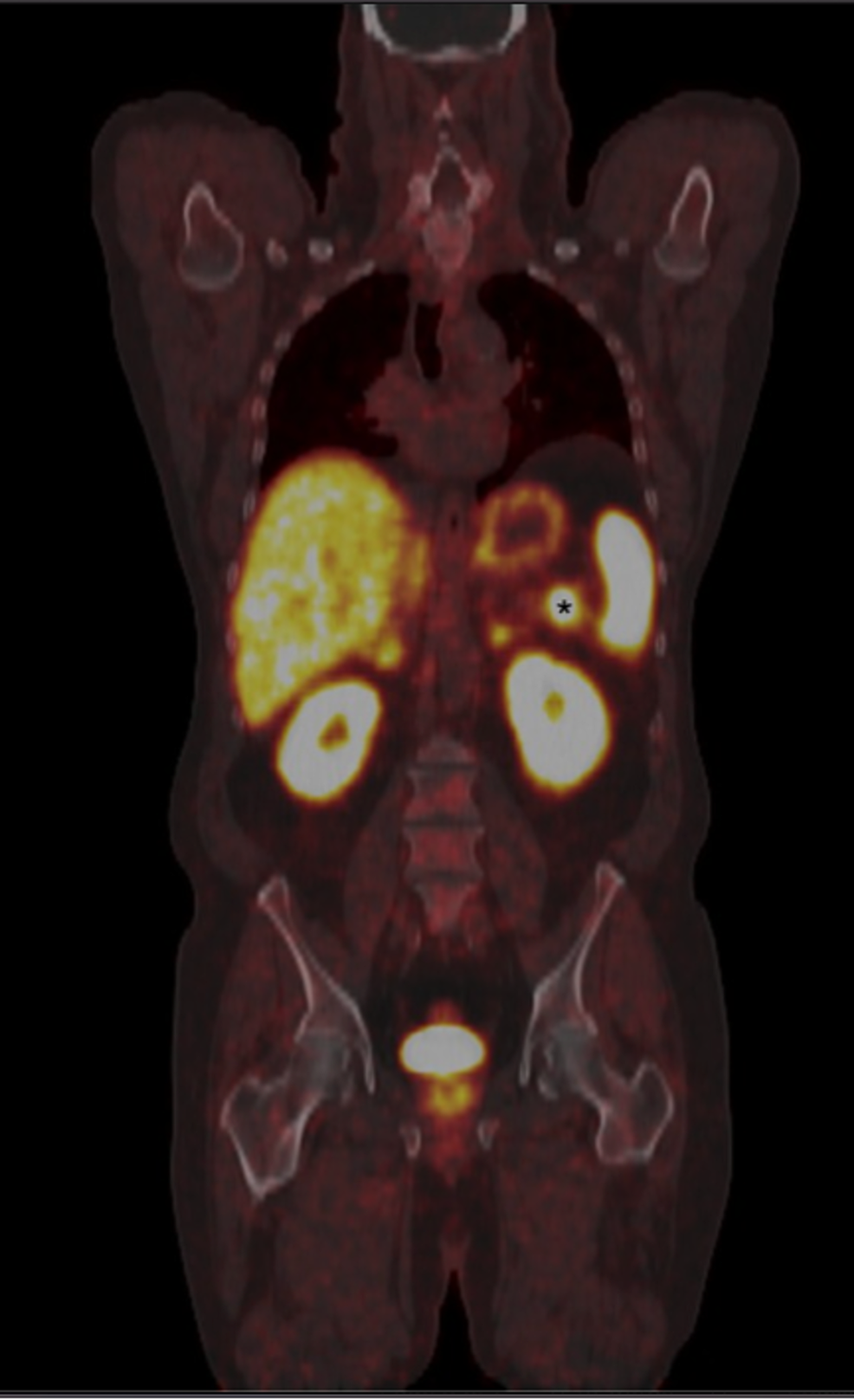
Food and Drug Administration (FDA) approved the use of gallium 68Ga-Dotatate, a radioactive diagnostic agent for positron emission tomography (PET) imaging to locate somostatin receptor positive neuroendocrine tumors in adult and pediatric patients.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
